Buscar
-
Managing Your Uncontrolled High Blood Pressure
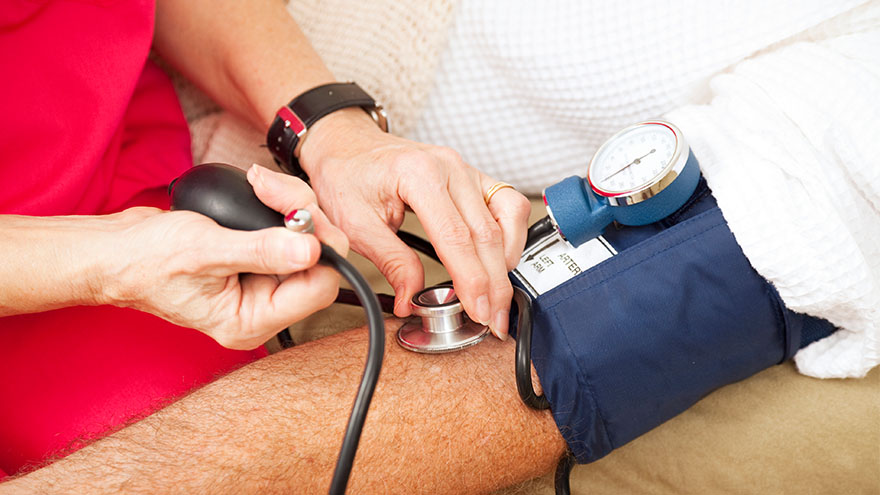
Renown Health, the region's leading cardiology care provider, is offering a clinical trial for eligible patients struggling to control their high blood pressure. Nearly half of adults (119.9 million) in the United States have hypertension, or blood pressure that is higher than normal. Hypertension can put you at risk of other life-threatening disease, such as a heart attack or stroke. There are methods that cardiologists use to manage high blood pressure, but only 1 in 4 adults with hypertension (27.0 million) have their blood pressure under control.* Some patients with high blood pressure experience resistant hypertension, which does not respond well to multiple antihypertensive medications given at the same time. This means that there are many important opportunities for healthcare professionals to explore new ways to treat hypertension. At Renown Health, we lead the region in cardiology care with our technological expertise and patient-centered approach. That is why our cardiology team is partnering with the Renown Research Office to offer the RADIANCE Continued Access Protocol (RADIENCE CAP) clinical trial to eligible patients. RADIANCE CAP is a non-randomized study designed to allow for continued access to ultrasound renal denervation therapy via the Paradise System, and to allow for the on-going collection of safety and effectiveness data in patients with uncontrolled hypertension despite the prescription of antihypertensive medications. The body’s complex communication system between the brain, heart and kidneys can sometimes become overactive, increasing your blood pressure through messages in the nervous system. Renal denervation is a minimally-invasive procedure which reduces activity from the nerves in your kidneys to lower blood pressure. This is the third in a series of renal denervation clinical trials Renown Health has offered to patients with resistant hypertension over the last several years, with over 40 local participants. “All the participants that I have had the pleasure to work with on these studies are very excited and grateful to have this option for helping control their blood pressure” states Lisa English, Lead Clinical Research Coordinator for Cardiology studies at Renown Health. “I love getting to know each one of them and helping on their healthcare journey. We have an amazing team of providers and staff at Renown that go out of their way to make patients experiences positive and the studies successful.” Dr. Michael Bloch, Cardiologist and Principal Investigator for the RADIANCE CAP study at Renown Health’s Institute for Heart and Vascular Health adds, “Despite lifestyle modifications like diet and exercise and the widespread availability of effective and well-tolerated medications, approximately 50% of all people with hypertension have inadequate blood pressure control putting them at risk for stroke, heart failure and kidney disease. As a one-time durable procedure, renal denervation with the Paradise endovascular system from ReCor Medical, Inc. may help millions of patients improve their blood pressure control without necessarily needing to increase their medications.” Our teams of expert providers and researchers are here to support you on your healthcare journey. Talk to your provider about the RADIANCE CAP clinical trial at your next appointment to see if participation may be right for you.
Read More About Managing Your Uncontrolled High Blood Pressure
-
Employee Blog: Team Renown at Northern Nevada Pride
It’s July 23, 2022, and I am arriving in Downtown Reno to walk in my first-ever Pride Parade. Even more amazing, I am going to be walking with a group of my coworkers, announcing to our entire community that our employer supports us being our full and truest selves. I arrived alone, but in the staging area I met new people who worked in other departments of Renown, coworkers I might never have met if not for Pride. I even took a selfie with one of these new friends. Someone passed around flags for us to hand out to the audience as we marched by. I took a rainbow-striped temporary tattoo in the shape of the Renown “R” and used my water bottle to apply it. There was an overwhelming atmosphere of excitement and joy that can only come from gathering with other human beings who are overflowing with love. We started the Parade waving handfuls of Pride flags high and gave those flags out to the audience as we passed. By the time we were done walking, our hands were empty, but our hearts were full. I couldn’t have stopped smiling if I’d tried. I’d joined the parade as a single person, but as we walked, I became part of a community. Not only the Renown community, but a community of Proud queer people across northern Nevada. And by extension, the LGBTQIA+ community all around the world – past, present and future. With the multiple recent state laws proposing to strip away the rights of LGBTQIA+ people, many of us have felt a noticeable decline in our mental health and feelings of personal safety. As a member of the queer community, I have felt these mental health effects too. I am fortunate and privileged enough to not have experienced workplace discrimination in my current position. But before Renown, I – like so many others in the LGBTQIA+ community – had a negative experience at a previous job, and afterwards I was hesitant to share my full self while at work. To be able to walk in a Pride Parade with my current employer, healed a little bit of that past pain inflicted by my former employer. When our liberties are under attack, Pride is more important than ever. We must remind ourselves, especially when others cannot seem to remember, that we are loved and worthy of love. I am grateful to work for an organization that is willing and able to fight this good fight with us. I will certainly be back to walk in this year’s Pride Parade, and I sincerely hope to see you there too! Be sure to wear comfortable walking shoes, sunglasses and of course keep your skin safe with sunscreen. Whether you’re part of the LGBTQIA+ community, or just an ally, you are welcome.
Read More About Employee Blog: Team Renown at Northern Nevada Pride
-
Top 5 Misconceptions About Clinical Trials
There are many misconceptions about clinical research, so we have unpacked a few common myths we hear to help you make an informed decision in your healthcare. Misconception #1: If I join a clinical trial, I’ll just be a guinea pig. Quite the opposite is true! Through honest and respectful conversation, we ensure all participants are informed of the benefits and risks associated with the clinical trial during the informed consent process. Being in a clinical trial is voluntary, and we respect our patients’ decision to join or decline to participate in the clinical trial. You can always change your mind at any time as well. When patients join a clinical trial, they receive an additional team of healthcare professionals, including additional physicians, pharmacists and research coordinators, dedicated to their safety and well-being. This means that clinical trial participants often receive more support than they would in the standard treatment setting. Misconception #2: Clinical trials are too dangerous because they use new treatments that haven’t been tested. We recognize that there are different levels of risk associated with participating in a clinical trial depending on the type of study. However, new treatments are only reviewed through clinical trials after they have gone through extensive testing. New treatments that do not show promising results for safety and potential benefit during laboratory testing do not receive approval to begin clinical trials. Your research team reviews any expected benefits and risks identified from previous studies during the informed consent process, as well as any updates that occur throughout the duration of the clinical trial. The research team stays in close contact with you during the entire process, documenting and treating any side effects that you experience for both your safety and the safety of participants like you. Misconception #3: I don't want to join a trial because I could be wasting my time receiving a placebo. A placebo is a substance that has no therapeutic effect, sometimes called a “sugar pill.” Participants who receive a placebo during a clinical trial are very important, helping researchers definitively determine the specific good and bad effects of the new medication. Many clinical trials that involve a placebo also offer what is called an open label extension or cross-over study. Cross-over studies ensure that anyone taking the placebo can begin receiving the new medication, often for several years. Cross-over studies help clinician researchers understand the long-term effects of a medication while also giving patients free access to novel care for several months and even years.
-
¿Qué significa participar en un estudio clínico?
Participating in a clinical trial is voluntary and a personal choice. Clinical trials are research studies that involve people and are an important part of patient care. What is a clinical trial? Clinical trials are research studies that involve people, and they are an important part of patient care. There are several different types of clinical trials; some are designed to understand trends in a disease or identify better ways to diagnose a condition, while others determine if a new treatment is safe and works when treating, improving or preventing a health condition. There are over 400,000 clinical trials currently being conducted in the United States, and even more across the world. This includes health conditions such as heart failure, cancer, Parkinson’s Disease, respiratory conditions like COPD, common infections, cystic fibrosis, and many more. Clinical trials lead the healthcare industry to new discoveries that contribute to reliable and exact care, improving healthcare quality and saving lives. Clinical trials are conducted by a team of researchers, including doctors, pharmacists and clinical research coordinators. These research teams are highly skilled in their specialty areas, often providing traditional patient care and seeing research patients in the same day. These teams are responsible for making sure the clinical trial is completed correctly, and their patients are their top priority. Why should I consider participating in a clinical trial? Participating in a clinical trial is voluntary and a personal choice. There are many reasons why patients decide to get involved in clinical research. While many clinical trials are designed for patients who have a certain health condition, many studies also ask healthy volunteers to contribute in order to compare health outcomes. Clinical trials are also for patients at all different stages of their diagnosis. Depending on the specific study, the patient may receive access to a new cutting-edge treatment before it is widely available. When patients join a clinical trial, the research team becomes a health partner dedicated to their health and well-being. When patients join a clinical trial, they make an informed decision in their healthcare by weighing all available options in addition to routine treatments. Research participants know that they are contributing meaningfully and helping other patients like them. Where can I find more information about clinical trials at Renown Health? Renown Health’s mission is to make a genuine difference in the health and well-being of the communities we serve. Renown’s clinical trial portfolio offers leading care options to patients in northern Nevada, close to home, in a variety of specialties. Contact the Renown Clinical Research Office for more information on clinical trials available to you!
Read More About What Does It Mean to Participate in a Clinical Trial?